Glycol Dehydration & Hydrate Prediction/Inhibition
ProMax is renowned as the leading resource for predicting the performance of both glycol dehydration and hydrate suppression systems.
ProMax can perform dehydration simulations using the common glycols Ethylene Glycol (EG), Diethylene Glycol (DEG), and Triethylene Glycol
(TEG). Dehydration processes using other solvents such as Methanol or DEPG can also be modeled. Injection systems using Methanol, EG, or other
hydrate inhibitors can be modeled as well. Almost any dehydration unit flow scheme may be simulated, including stripping gas, stripping gas
with dryer column, solvent enhanced stripping, and enhanced stripping with vapor recovery. For cases involving Methanol, a special Polar Equation
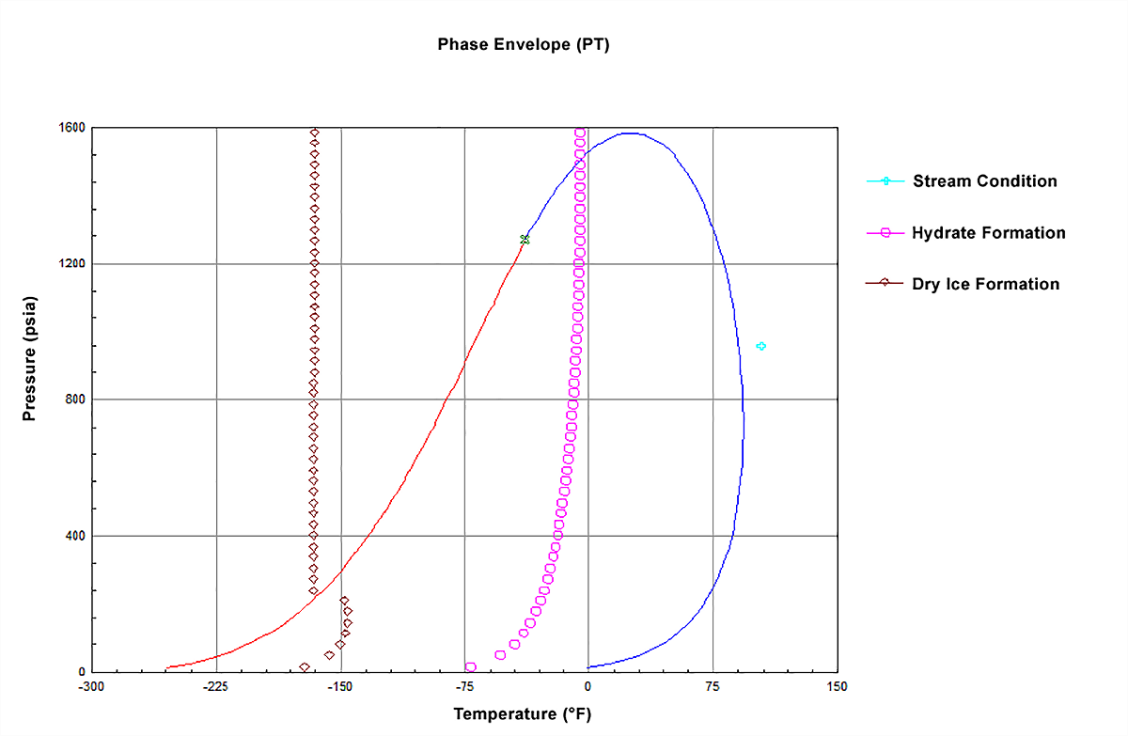
of State Property Packages is available. A special Freeze-Out Analysis automatically calculates the highest formation temperature for ice,
solid CO2, and hydrates as well as the possibility for multiple formation temperatures of each type. In addition, it calculates water dew point
and water content which are very useful properties for dehydration applications.
Hydrate Inhibition
-
Model injection systems using Methanol or glycol
-
Inhibition utilizes injection of one of the glycols or methanol into a process stream where it can combine with the condensed aqueous phase
to lower the hydrate formation temperature at a given pressure.
-
Both glycol and methanol can be recovered with the aqueous phase, regenerated, and reinjected.
-
Accurate Methanol-Water-Hydrocarbon VLE/VLLE/LLE predictions
Stripping Gas
-
A portion of the sales gas (or other available gas) is injected directly into the reboiler to aid in stripping.
Stripping Gas with Dryer (Stahl Column)
-
A glycol stripper with a dryer can achieve a more pure glycol than stripping gas injected
directly into the reboiler since the dryer provides additional ideal stages for stripping.
-
A dryer is a secondary column attached below the stripper reboiler which further reduces the water content of the lean glycol. It is usually a small
packed column with no more than one or two ideal stages. The stripping gas is injected in the bottom stage of the dryer.
Solvent Enhanced Stripping
-
A volatile hydrocarbon liquid is fed into the glycol regeneration system to increase the volatility of water in the solution and, after vaporization,
act as stripping gas in the regenerator. Mixed vapors flow through the glycol reboiler and the rich glycol stripper, which is refluxed with aqueous
condensate to minimize glycol losses. Hydrocarbon vapors from the rich glycol stripper are condensed and recycled to the regeneration system. Liquid water
is discarded. Dew points near -150F can be achieved with this technology.
-
The DRIZO® technology is an example of process that uses solvent enhanced stripping. The vapor from the glycol reboiler/still column is condensed in a
unit together with the vapor from the stripping column. The solvent and all BTEX compounds are condensed in this unit before the vapor is discharged to
atmosphere. The main advantages of this system are that all BTEX compounds are recovered from the vapor before being sent to atmosphere. The TEG purity
can be as high as 99.999 wt%, and no external stripping gas is required. The DRIZO technology may be adapted to existing units which need to be upgraded
due to new requirements for higher glycol purity, or for better emission control of BTEX and CO2.
Coldfinger®
-
The Coldfinger process is based on a conventional glycol regeneration unit. The main principle of this technology is to condense and collect
water/hydrocarbons from the reboiler vapor phase and drain it away from the reboiler.
-
A cooling coil (the Coldfinger condenser) is placed in the vapor space above hot glycol from the reboiler of a conventional regenerator, and a collecting tray
is placed below the coil to catch condensate. Since the vapor above concentrated glycol is much richer in water vapor than in glycol vapor,
its condensation removes water from the system, and additional water is vaporized from the hot glycol.
Adsorber (solid desiccant/molecular sieve) Dehydration
- Dehydration by adsorption uses an adsorber packed with solid desiccant or a molecular sieve to reduce the water content of wet gas.
- ProMax utilizes the GPSA calculations for sizing an adsorber to calculate:
- Adsorption Cycle Time
- Adsorber Pressure Drop
- Adsorber Inside Diameter
- Adsorber Height
- Dehydration adsorbers continue to adsorb water until they are saturated and then must be regenerated through stripping gas or heat.
- ProMax calculates the heat required for regeneration as well as the:
- Regeneration Cycle Time
- Regenerator Pressure Drop
- Mass Flow of Regeneration Gas